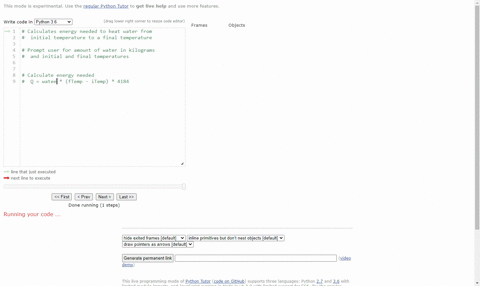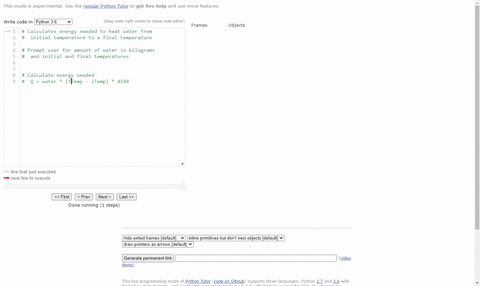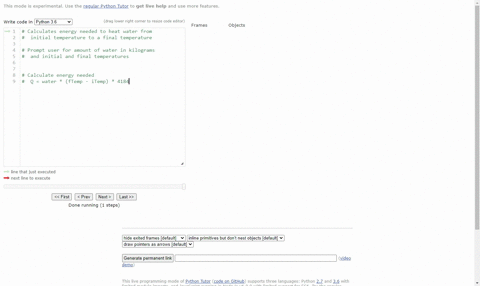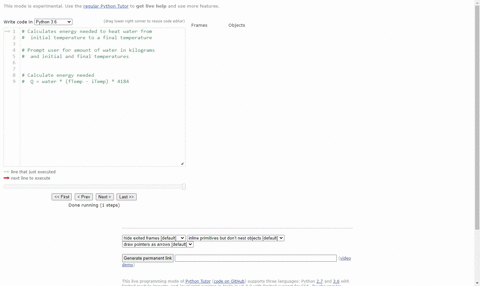
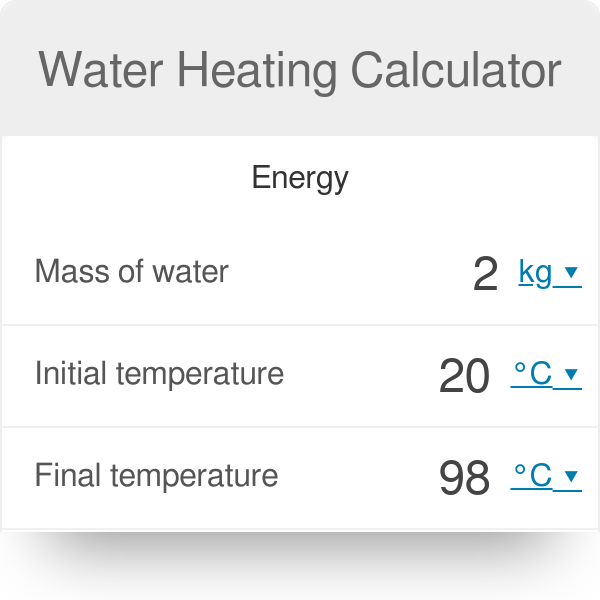
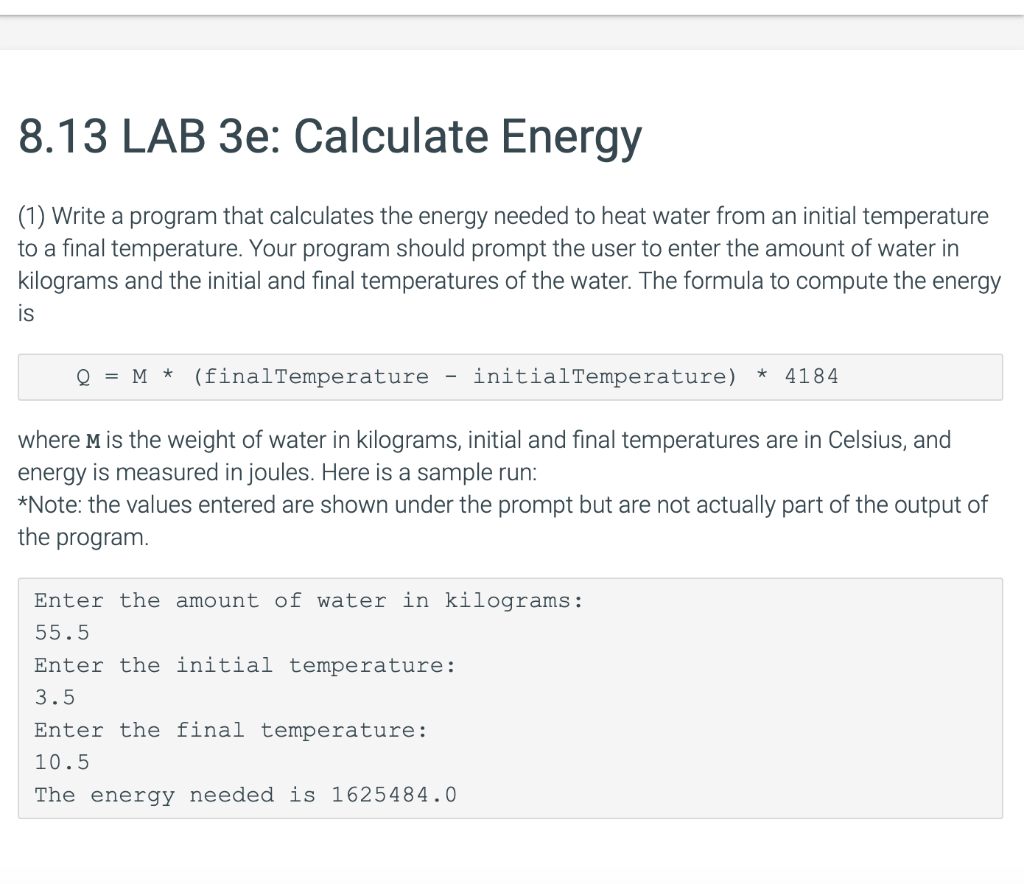
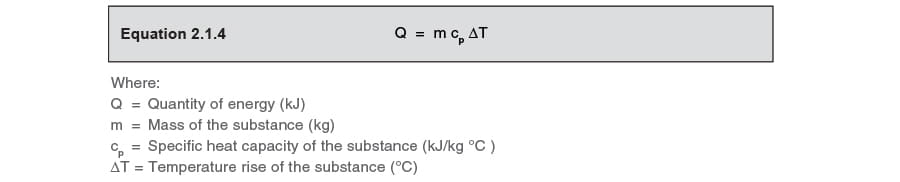
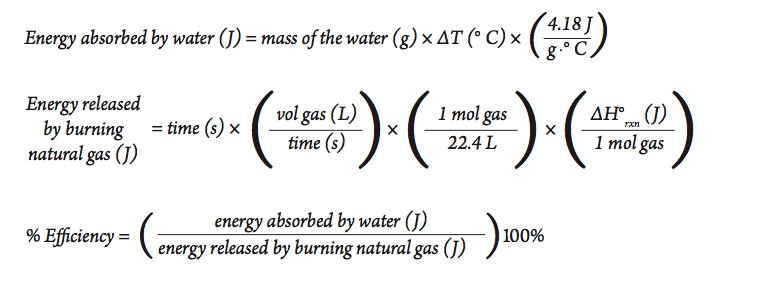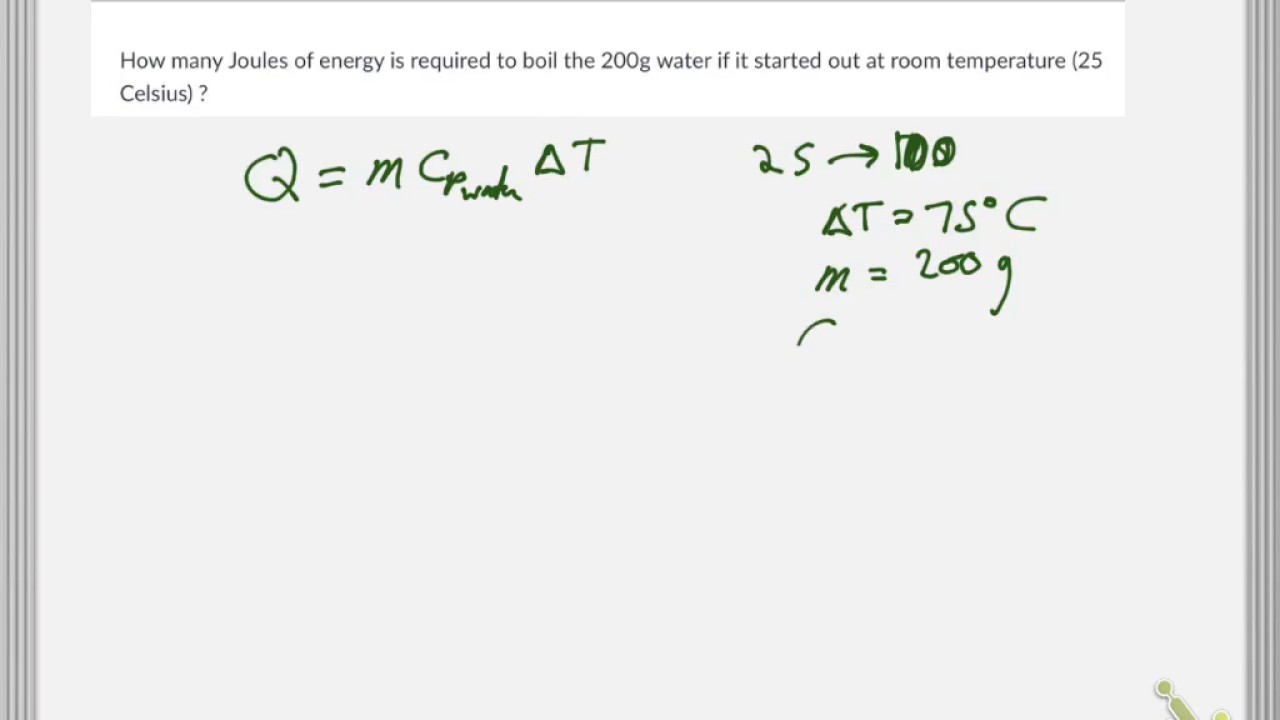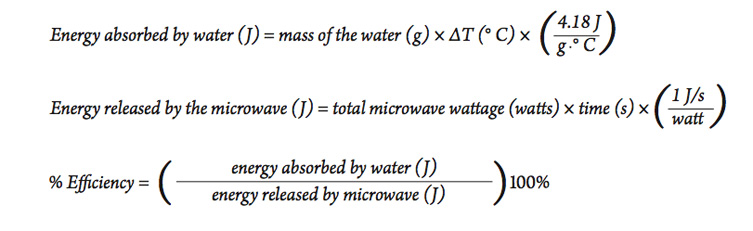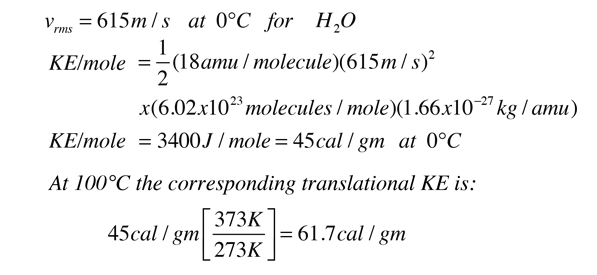Calculate the heat energy required to raise the temperature of 2 kg of water from 10^@C to 50^@C. Specific heat capacity of water is 4200 JKg^(-1) K^(-1)

Calculating Heat. Specific Heat Amount of heat energy needed to raise the temp of 1 ml of a substance 1°C For water the specific heat is 4.19 J/g °C, - ppt download

The heat energy required to raise the temperature of 2kg of water from 10^0C to 50^0C is (Specific heat capacity of water is 4200 J Kg^0C^-1)

Enthalpy. Specific Heat Capacity Definition: The HEAT ENERGY required to raise the TEMPERATURE of 1kg of substance by 1 o C. e.g. for water C= 4.18kJ. - ppt download

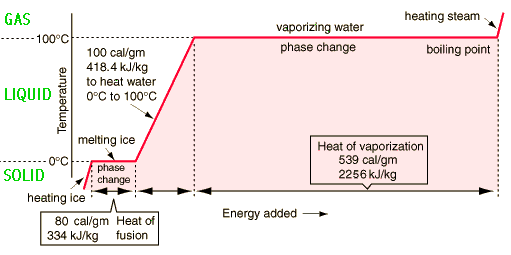
Question Video: Finding the Amount of Energy Needed to Change the State of Water from Liquid to Gas | Nagwa
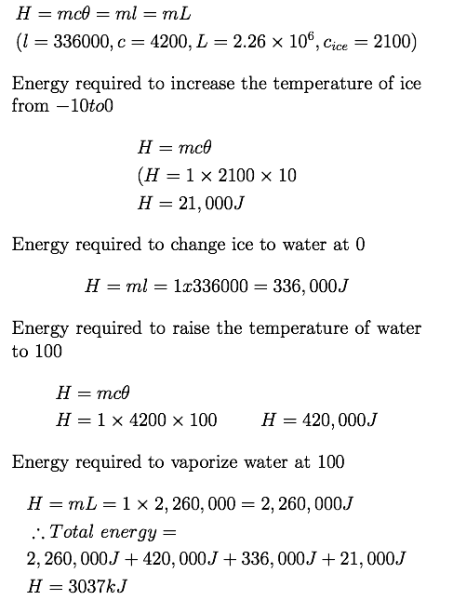
calculate the total energy required to evaporate completely 1kg of ice that is initially at... - Myschool

Calorie (energy) Calculations A calorie is defined as the amount of energy it takes to raise the temperature of one gram of water by one degree Celsius. - ppt download

Heat and Temperature Heat is a form of energy, and is measured in Joules (J). Temperature is different from heat. Temperature is a measure of how hot or. - ppt video online