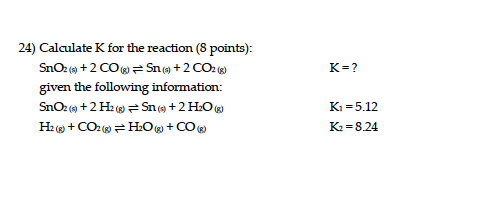Calculate the equilibrium constant for the the reaction given below at 400K, if Delta H ^(@) = 77.2 kJ mol ^(-1) and Delta S ^(@) = 122JK ^(-1) mol ^(-1) , PCl (s (s)) to PCl (3 (g)) + Cl (2 (g))

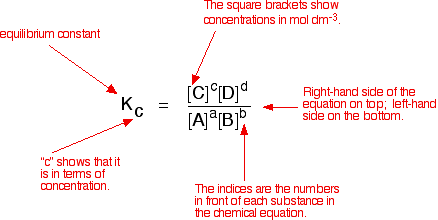
The value of Kc = 4.24 at 800 K for the reaction CO(g) + H2O(g) CO2(g) + H2(g) Calculate equilibrium concentrations of CO2, H2, CO and H2O at 800 K , if
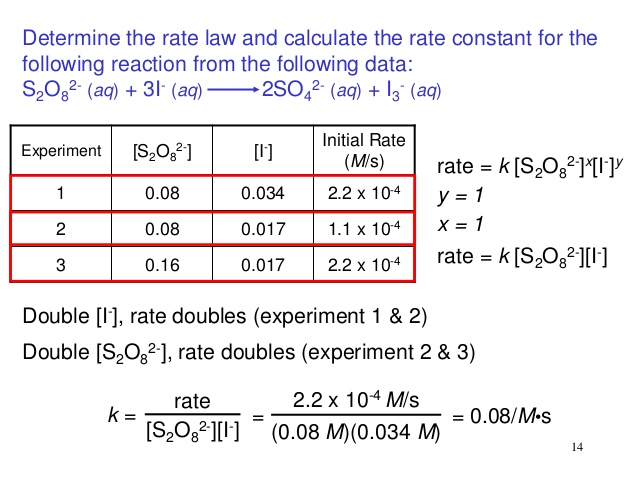
Is it true that the value of k depends on the amount of reactants and products mixed together initially? | Socratic

Sample Exercise 15.7 Calculating K When All Equilibrium Concentrations Are Known After a mixture of hydrogen and nitrogen gases in a reaction vessel is. - ppt video online download

Calculate the equation constant for the reaction: H2 (g) + CO2 (g) H2O (g) + CO at 1395 K, if the equilibrium constants at 1395 K for the following are 2H2O (g)



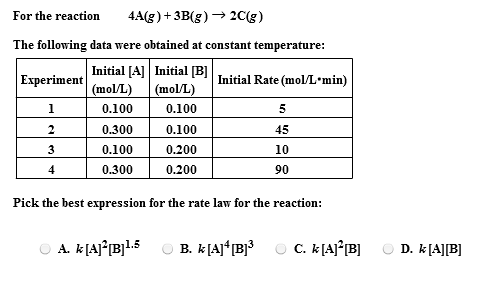

![Calculate K from [EQUILIBRIUM] concentrations 2018 - YouTube Calculate K from [EQUILIBRIUM] concentrations 2018 - YouTube](https://i.ytimg.com/vi/8l1HCaVLgVI/maxresdefault.jpg)