SOLVED: Use the data provided below to calculate the lattice energy of RbCL Is this value greater or less than the lattice energy of NaCl? Explain Electron affinity of Cl = -349
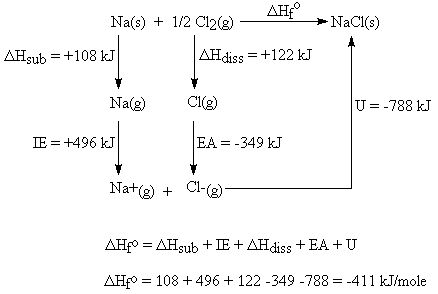
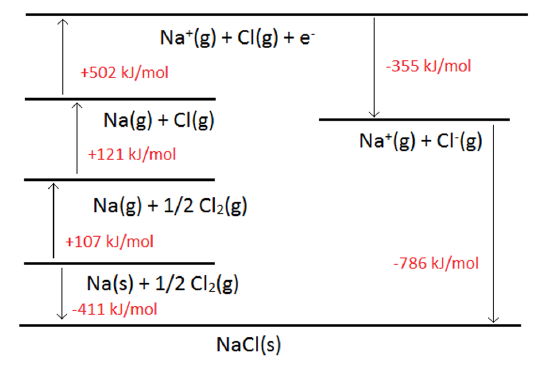
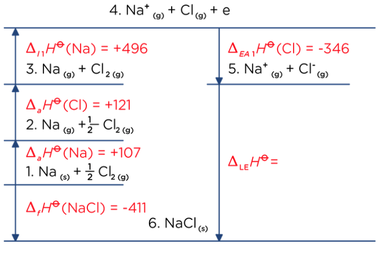
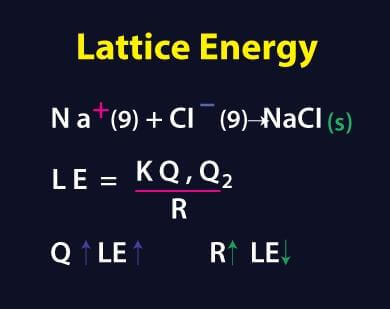
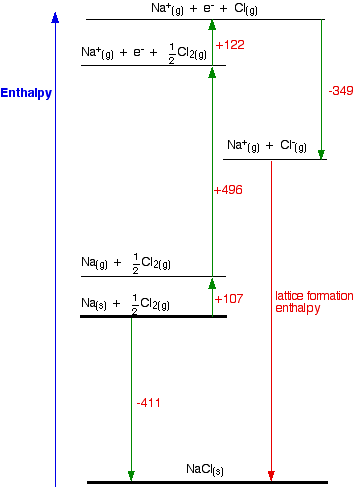
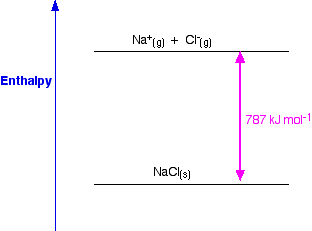
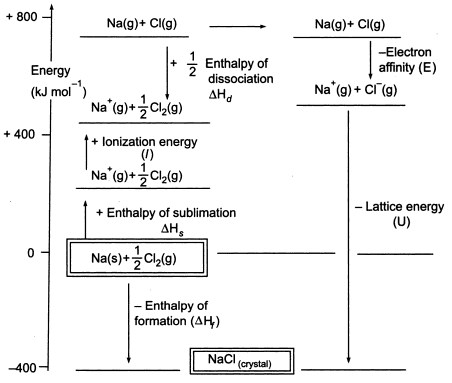
What is lattice enthalpy? How do you calculate the lattice enthalpy of sodium chloride by using Born-Haber's cycle?

SOLVED:Calculate the lattice energy of sodium oxide (Na2 O) from the following data: Ionization energy of Na(g)=495 kJ / mol Electron affinity of O(g) for 2 electrons =603 kJ / mol Energy

How do you calculate the lattice energy of NaF using the Born-Haber cycle? (Delta Hsub for sodium is 107.5 kJ/mol) | Homework.Study.com

Born Haber cycle/Calculation of lattice energy for sodium chloride/ Hess's law/ Thermodynamics - YouTube

Born Haber Cycle, Basic Introduction, Lattice Energy, Hess Law & Enthalpy of Formation - Chemistry - YouTube